![]()
Take the Genesis Biosciences tour
Explore our state-of-the-art facility & learn more about our unrivalled staff expertise
Our equipment is industry-leading
Custom engineered, high-capacity fermenters housed within a well-planned newly rebuilt facility power our microbial production, allowing for enhanced process efficiency and extreme cleanliness that ensures each fermentation result is stable and pure. Compartmentalization and enclosure of the upstream processing equipment eliminates risks of outside environmental contamination and improves safety and security. Purposely designed manual control overrides are in place to enable continued production if automation fails ensuring high levels of success when producing our or your strains.
Our fermenters along with our fermentation expertise enable us to successfully scale up from bench top pilot fermentations to a range of larger vessels to meet the varying needs of our customers.
For optimized downstream processing, our state-of-the-art spray dryer, fully contained centrifuge, and other downstream processing equipment are strategically placed in a fully separate, sanitized suite to meet compliance requirements. Our unique and proprietary CIP procedures focus on absolute sterility so you can be assured of the highest quality for our methods of production.
"We've made significant capital investment in our company and staff, which started with big improvements to our facility last year. We're continually striving to lead the way using better technologies within our industry."
Conrad Mielcuszny, CEO of Genesis Biosciences
Ensuring quality as standard in our workplace
With brand new antimicrobial flooring fitted in July 2018, our facility provides maximum safety and hygiene in fermentation, production, downstream processing and the warehouse areas.
Our previous lab expansion has supported our infrastructure for growth with the addition of two new labs that give us dedicated areas for additional research and development, three office suites for our technical staff and a new retention sample room.
All the walls and ceilings in our facility are upgraded with non-porous cleanable surfaces for improved sanitization.
HVAC throughout our entire business delivers complete climate control for increased staff comfort, maximum energy efficiency and decreased environmental risks.
- Brand new antimicrobial flooring
- Maximum safety and hygiene in fermentation, production, downstream processing and the warehouse areas
- Two new labs with dedicated areas for additional research and development
- Three office suites for our technical staff
- New retention sample room
- Upgraded walls and ceilings with non-porous cleanable surfaces for improved sanitization
- HVAC throughout our entire business delivers complete climate control
Ensuring quality in our process and systems is paramount
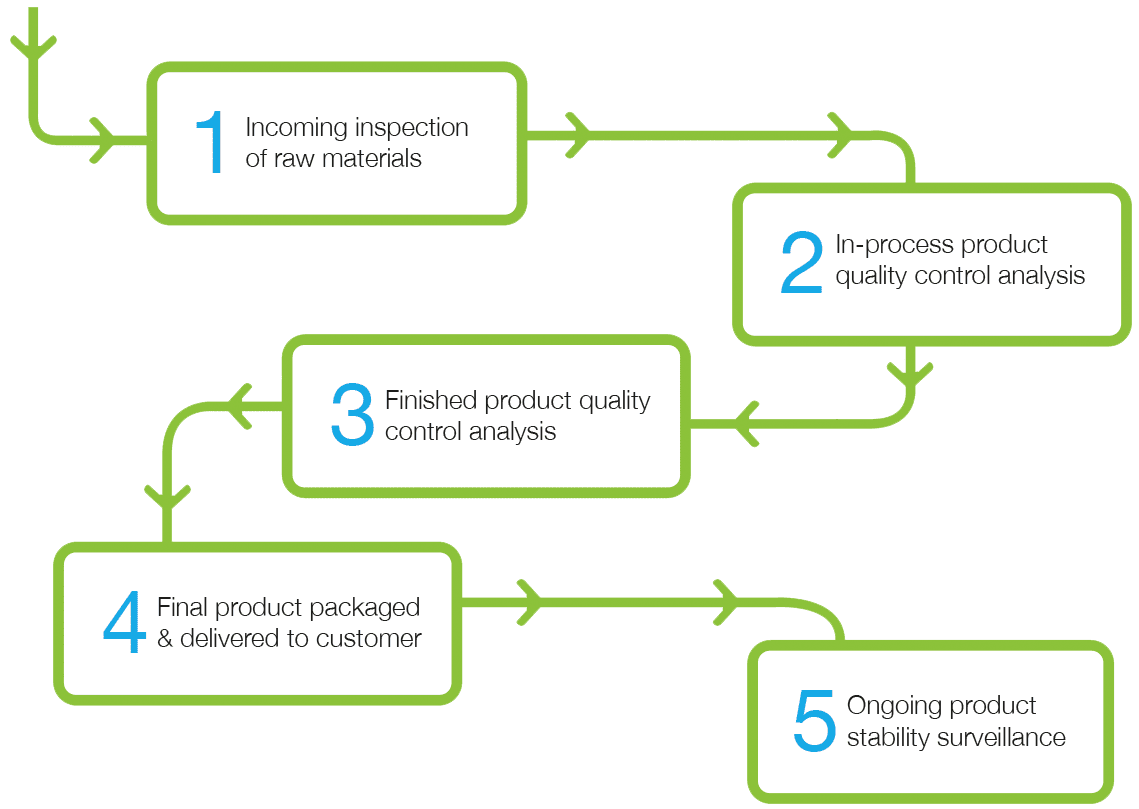
Genesis Biosciences applies stringent Quality Control (QC) checks at all stages of the production process, from incoming inspection of raw materials, in-process and finished product quality control analysis to ongoing product stability surveillance. Our in-depth QC screening processes ensure that all products meet our rigorous quality specifications.
In depth validation at critical control points throughout the manufacturing process allow close monitoring of spore count and ensures the absence of environmental contaminates from Cryotube to finished product.
Genesis Biosciences’ Compliance Taskforce is made up of highly experienced Quality Assurance and Quality Control personnel with extensive chemistry, fermentation and FDA inspection/auditing expertise. This ensures monitoring and preventative controls are implemented at critical control points throughout the fermentation and production processes in order to yield the highest quality product possible.
Our optimized processes deliver efficiency and the highest quality for our methods of production as we adhere to cGMP, ISO22000, HACCP and FSMA compliance.

Debbie Williams
Group Compliance Officer
"Working at Genesis Biosciences means being part of something bigger. I come to work every day at a place where the core values match my own, and I can feel proud in my career."
Stephanie Vazquez, Sales Administrator
Explore our facility floorplan
It's our team that makes us special
Find out what makes our company unique
From our state-of-the-art fermentation equipment to our membership of the industry's most prestigious trade associations, find out how we're committed to leading the way in scientific advancement.

Laboratory equipment
- Laminar Flow Hood
- Spectrophotometer
- BSL-2 Biological Hoods
- Homogenizers
- Biological Safety Cabinet
- Autoclaves
- Microscope
- FOG Analyzer
- Sub-zero freezer
- Microplate Reader
- Labware washer with DI rinse

Fermentation equipment
- Fermentors: Benchtop/ 500L / 2000L / 4000L / 30000L
- pH and Foam Control Tanks
- Autoclaves
- 1600L Batch tank
- Centrifuge
- Spray Dryer (100 kg/hour)
- Air Scrubber System
- Dust Collection System
- Chill water systems
- Steam Generators
- Pressure/Air Generators
- Parameter Controls (ph, temperature, dissolved oxygen)
- Holding tanks

Production equipment
- Liquid Batch Tanks: 220 Gallon / 1200 Gallon / 660 Gallon (x2) / 220 Gallon (x2) / 110 Gallon / 20 Gallon
- Ribbon Blenders: 100 cft / 60 cft (x2) / 10 cft / 6 cft
- Vertical Form Fill & Seal equipment
Trade Associations





Find out more about our ethos & commitment to quality
Our product ranges

The Evogen range provides high-quality, performance-driven, environmentally-responsible, solution-specific microbial products offering effective and sustainable alternatives to traditional chemical and microbial products across a wide range of industries

The Evobac range is a series of performance-driven biological broth formulations for use across a wide range of industries - from aquaculture to wastewater treatment and beyond.

The Evozyme range is a series of microbial formulations for use across a wide range of industries - from all-purpose cleaner to pond and lake management and beyond
Latest facility updates

Brand new flooring
We recently fitted brand new antimicrobial flooring in our facility - watch our video to see it transform in under 2 minutes.

Facility improvements
We're committed to investing in our facility, our people, and our future - find out how we're doing it.

Meet the experts
Our newly-strengthened core team bring over a century's experience and expertise - meet the Genesis team.







